On April 13th, the administration of Johnson and Johnson’s Covid-19 vaccine was paused after six people – out of the 6.8 million people who had taken the vaccine – developed a rare blood-clotting condition called cerebral venous sinus thrombosis (CVST). In general, this condition is rare, affecting five people per million every year.
After the CDC and the FDA made this recommendation out of an “abundance of caution,” there has been a lot of coverage focusing on the risk of blood clots in general. Many have pointed out that there is a much higher risk of getting a blood clot from birth control pills, smoking a cigarette, or Covid-19 itself, than from the Johnson and Johnson vaccine.
This is true, but it isn’t a fair comparison because the blood clots linked to birth control are different from those linked to this vaccine.
CVST is particularly rare and occurs when a blood clot forms in the area of the brain known as the venous sinus and stops blood from draining out of the brain, which can lead to a stroke. In the majority of strokes, blood clots form in the arteries, not the brain. The blood clots linked to birth control also don’t form in the brain – but rather in a deep vein including the leg or pelvis.
The important question here is:
How does the risk of CVST, specifically, from taking the Johnson & Johnson COVID vaccine compare to other commonly prescribed medications in the U.S?
We examined data from the FDA Adverse Events Reporting System, looking at all the reports that potentially tied a medication to cerebral venous sinus thrombosis.
We found that not only is developing CVST blood clots are rare for all prescribed medications but that the Johnson & Johnson vaccine is among medications with the lowest risk.
In 2018, over 650 different drugs were associated with cerebral venous sinus thrombosis in at least one patient. Just as with the vaccine cases, these are reports submitted that potentially tie a medication to an adverse reaction. It’s not guaranteed that these medications did cause the CVST, just that they were being taken by the patient at the time of the adverse reaction.
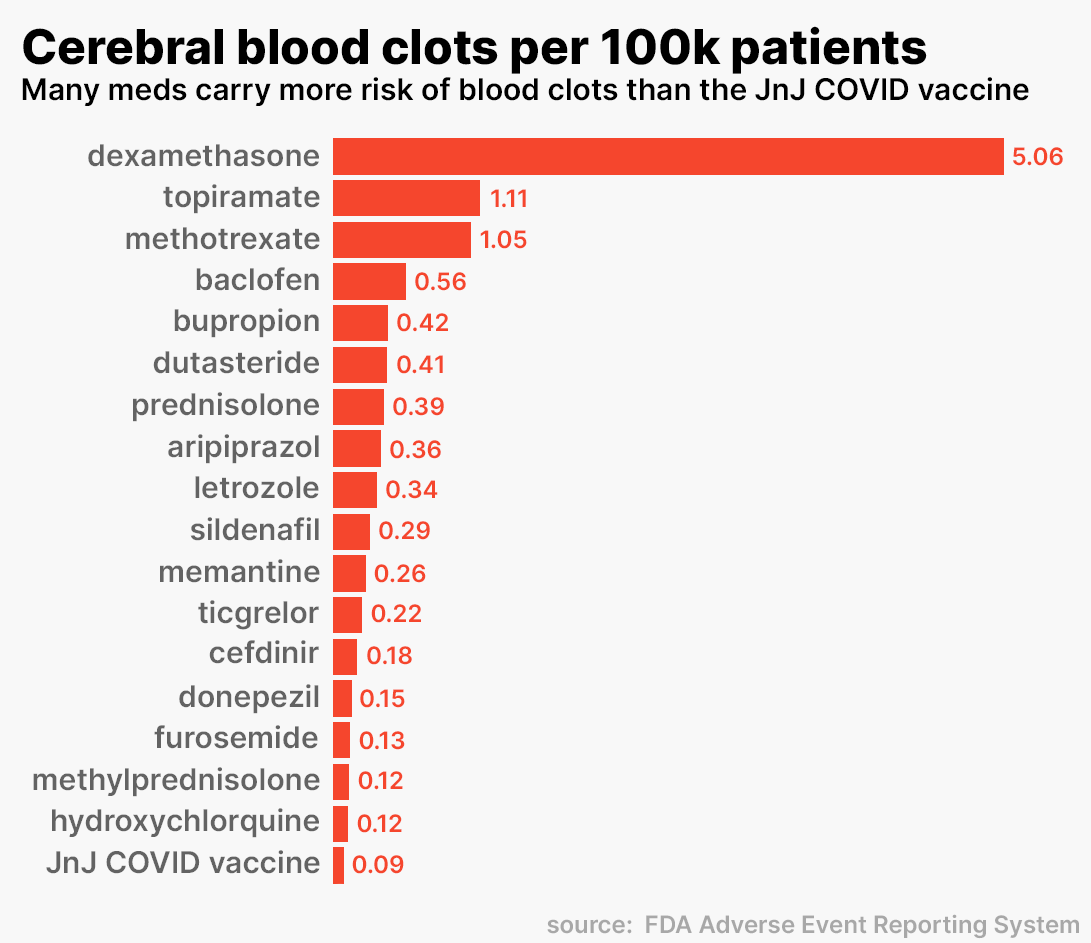
Only 17 of the 650 were associated with eight or more CVST blood clots and all 17 had a prescription adjusted rate higher than that of the Johnson & Johnson vaccine.
17 Commonly Prescribed Drugs Associated with CVST
| Drug Name | Brand Name | Treats | CVST in 2018 | Rx in 2018 | CVST per 1m |
|---|---|---|---|---|---|
| Dexamethasone | Ozurdex, Maxidex | Inflammation | 215 | 4,252,710 | 50.56 |
| Topiramate | Trokendi XR, Qudexy XR, Topamax | Seizures | 96 | 8,648,513 | 11.10 |
| Methotrexate | Rasuvo, Otrexup (PF), Xatmep | Cancer | 173 | 16,554,738 | 10.45 |
| Baclofen | Gablofen, Lioresal | Muscle pain | 63 | 11,285,060 | 5.58 |
| Bupropion | Wellbutrin | Depression | 102 | 24,488,843 | 4.17 |
| Dutasteride | Avodart | Urinary retention | 11 | 2,674,840 | 4.11 |
| Prednisolone | red Mild, Pediapred, Omnipred | Inflammation | 61 | 15,607,800 | 3.91 |
| Aripiprazole | Abilify | Schizophrenia, bipolar disorder, depression | 27 | 7,406,248 | 3.65 |
| Letrozole | Femara | Breast cancer | 16 | 4,755,100 | 3.36 |
| Sildenafil | Viagra | Erectile dysfunction | 16 | 5,565,826 | 2.87 |
| Memantine Hydrochloride | Namenda | Dementia | 8 | 3,129,750 | 2.56 |
| Ticagrelor | Brilinta | Acute coronary syndrome | 8 | 3,587,020 | 2.23 |
| Cefdinir | Omnicef | Bacterial infection | 8 | 4,361,025 | 1.83 |
| Donepezil Hydrochloride | Aricept | Dementia | 8 | 5,412,435 | 1.48 |
| Furosemide | Lasix | Fluid retention | 119 | 89,573,835 | 1.33 |
| Methylprednisolone | Depo-Medrol, Medrol, Solu-Medrol | Inflammation | 15 | 12,137,886 | 1.24 |
| Hydroxychloroquine | Plaquenil | Lupus, malaria | 13 | 10,812,380 | 1.20 |
What Does This Mean?
Which medication seems to have the highest risk of CVST? Dexamethasone, a steroid used to treat inflammation, had the highest rate of blood clots reported to the FDA (5.0556 cases per 100,000). The Johnson & Johnson vaccine, on the other hand, had a rate of .09 per 100,000 prescriptions.
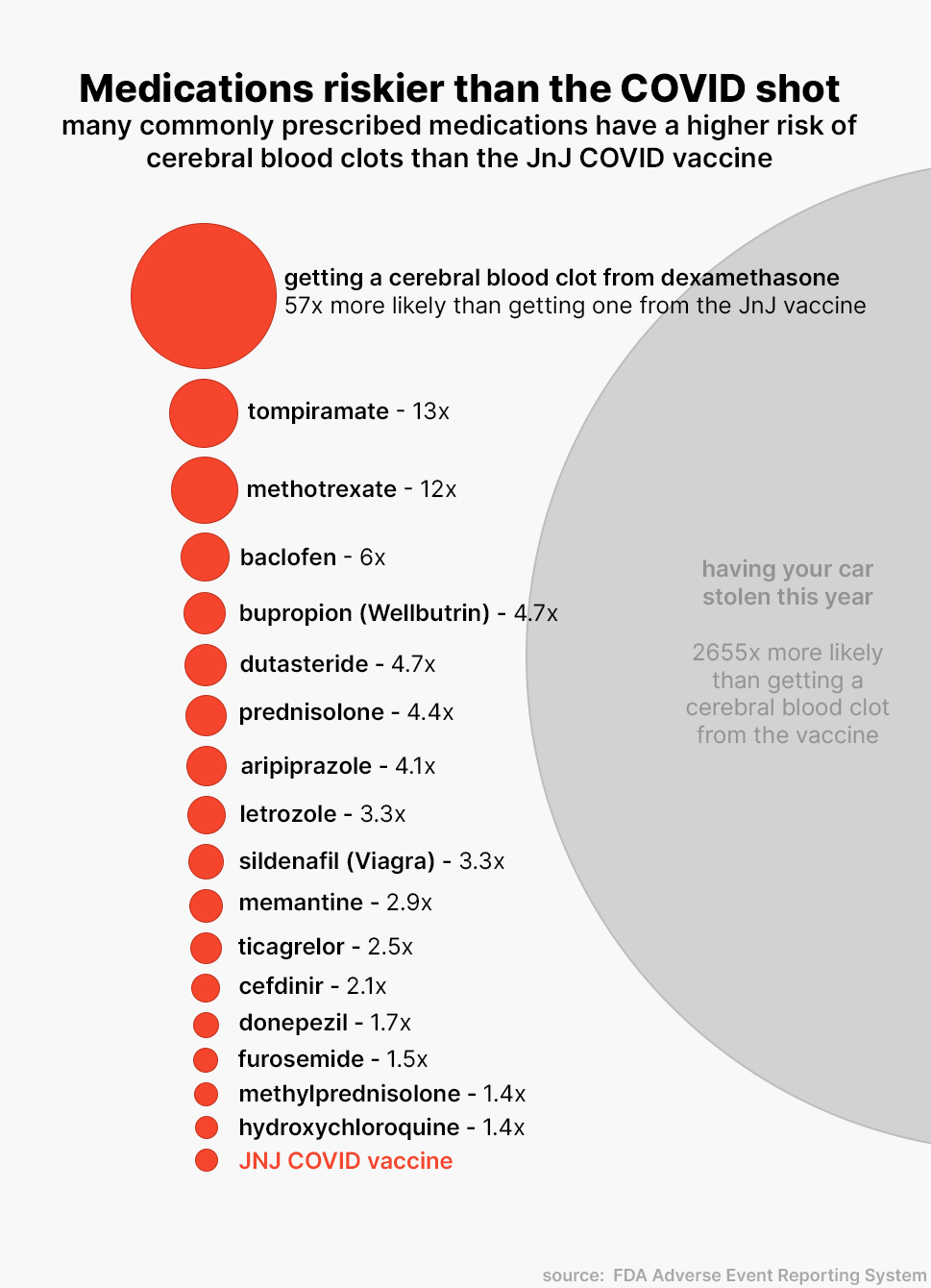
This means that patients were 57 times more likely to get a cerebral blood clot from Dexamethasone than from the Johnson & Johnson vaccine. In fact, you are much more likely to develop a CVST blood clot by treating erectile dysfunction. Viagra, a medication familiar to most, carries a risk 3.3 times higher than the vaccine.
Essentially, the risk of developing a CVST blood clot from any medication is low. The risk from receiving the Johnson & Johnson vaccine specifically is minuscule. To put it into perspective, you are 2,655 times more likely to have your car stolen this year.
Data Sources and Takeaways
This study emphasizes that the risks for CVST are extremely rare for both prescription medications and the vaccine, and the benefits far outweigh the risks.
The 2018 data used in our analysis comes from the FDA Adverse Event Reporting System (FAERS). These medications were taken at the same time the patient experienced the adverse reaction, but we cannot pinpoint whether they were the only cause of CVST.



No Comment